-
Description
-
Content
-
Reflection
<
>
For this project, we had to design a board game that incorporates chemical reactions. We had to include a single replacement reaction, double replacement reaction, a production of a gas, and light an LED. We presented our games at our Family Board Game night.
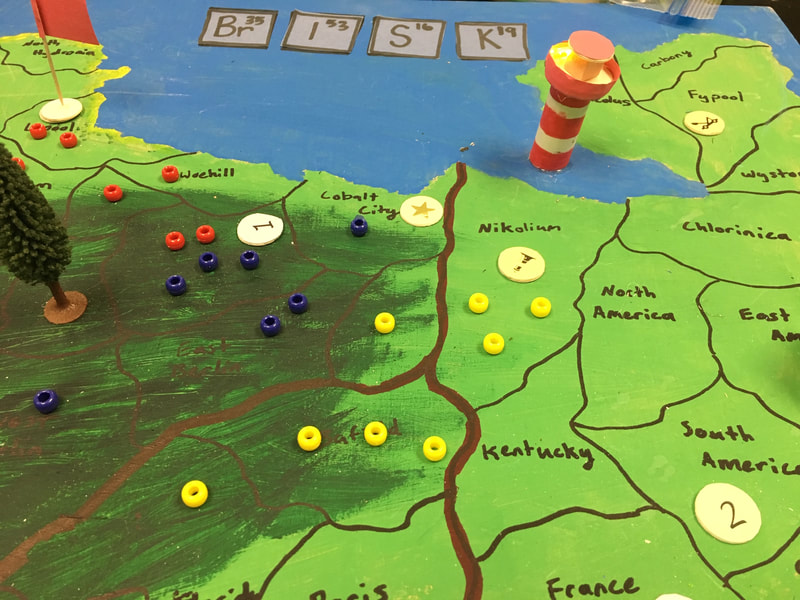
My team created a game similar to Risk called, BrISK. Your goal was is to capture territories with chemical reactions in them. When you have control of 4 chemical reactions you win the game. Our LED is in our lighthouse and a reaction launches a catapult by filling a balloon up with gas. You can read more about each reaction in our game below.
My team created a game similar to Risk called, BrISK. Your goal was is to capture territories with chemical reactions in them. When you have control of 4 chemical reactions you win the game. Our LED is in our lighthouse and a reaction launches a catapult by filling a balloon up with gas. You can read more about each reaction in our game below.
Single Replacement Reaction:
This is also called a Single Displacement reaction. A reaction where an element and a compound combine and the element replaces one of the elements in the solution. That element forms a compound with the other element and the element that used to be in the compound is now just an element. For a visual representation see slide 3 in our slideshow. A single replacement reaction in our game was reaction 1. We combined Steel Wool and Copper Chloride which yields Copper and Iron Chloride.
Double Replacement Reaction:
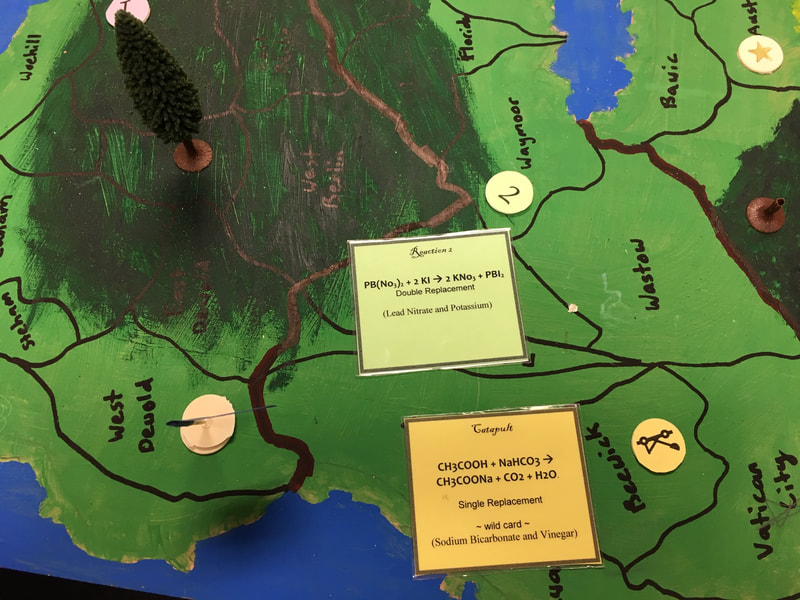
This is also called a Double Displacement reaction. A reaction where two compounds combine and an element in each compound switches to form a compound with the other element's pair. For a visual representation see slide 3 in our slideshow. This creates two new compounds. In our game, a double replacement example is reaction 2. We mixed Lead Nitrate and Potassium Iodide to yield Potassium Nitrate and Lead Iodide.
Synthesis Reaction: a reaction where two elements or compounds combine to form one compound. we did not have any of these in our game.
Decomposition Reaction: a reaction where one compound breaks apart into several elements or compounds. We did not have any of these in our game.
Combustion Reaction: a reaction where a combustible compound reacts with an oxidizer. We did not want to use fire or heat in our board game so we don't have any combustion reactions.
Reactant: An element or compound that is used in a reaction. We used reactants for each of our reactions.
Product: An element or compound formed after a reaction. The products are what make it so you could see our reactions. A product of Carbon dioxide filled our balloon.
Element: a substance formed by atoms that is a building block for everything. We used many different elements in our reactions.
Compound: A substance formed by different types of elements combining. We used several compounds in our game.
Activity Series:
An order of elements based on their reactivity which determines which elements will react with each other and how reactive they will be. We used this to predict which reaction would work.
Chemical Equation: a way to write an equation that shows the quantity of reactants and products. When balanced, they take into account the charges of the ions. We wrote one for each of our reactions.
Ion: an atom with a charge. We used them in our chemical equations.
Proton: a positively charged subatomic particle. They are in the nucleus. They are in all matter.
Neutron: a neutral subatomic particle. They are in the nucleus. They are in all matter.
Electron: a negatively charged subatomic particle. They are in the electron cloud. They are in all matter.
This is also called a Single Displacement reaction. A reaction where an element and a compound combine and the element replaces one of the elements in the solution. That element forms a compound with the other element and the element that used to be in the compound is now just an element. For a visual representation see slide 3 in our slideshow. A single replacement reaction in our game was reaction 1. We combined Steel Wool and Copper Chloride which yields Copper and Iron Chloride.
Double Replacement Reaction:
This is also called a Double Displacement reaction. A reaction where two compounds combine and an element in each compound switches to form a compound with the other element's pair. For a visual representation see slide 3 in our slideshow. This creates two new compounds. In our game, a double replacement example is reaction 2. We mixed Lead Nitrate and Potassium Iodide to yield Potassium Nitrate and Lead Iodide.
Synthesis Reaction: a reaction where two elements or compounds combine to form one compound. we did not have any of these in our game.
Decomposition Reaction: a reaction where one compound breaks apart into several elements or compounds. We did not have any of these in our game.
Combustion Reaction: a reaction where a combustible compound reacts with an oxidizer. We did not want to use fire or heat in our board game so we don't have any combustion reactions.
Reactant: An element or compound that is used in a reaction. We used reactants for each of our reactions.
Product: An element or compound formed after a reaction. The products are what make it so you could see our reactions. A product of Carbon dioxide filled our balloon.
Element: a substance formed by atoms that is a building block for everything. We used many different elements in our reactions.
Compound: A substance formed by different types of elements combining. We used several compounds in our game.
Activity Series:
An order of elements based on their reactivity which determines which elements will react with each other and how reactive they will be. We used this to predict which reaction would work.
Chemical Equation: a way to write an equation that shows the quantity of reactants and products. When balanced, they take into account the charges of the ions. We wrote one for each of our reactions.
Ion: an atom with a charge. We used them in our chemical equations.
Proton: a positively charged subatomic particle. They are in the nucleus. They are in all matter.
Neutron: a neutral subatomic particle. They are in the nucleus. They are in all matter.
Electron: a negatively charged subatomic particle. They are in the electron cloud. They are in all matter.
This project was very frustrating. We attempted to be innovative and create a domination board game instead of a Candyland style game. However, this means that we did not have a domino effect which was a requirement for the project. We did not use the first few work days as effectively as possible. Additionally, the team dynamics were very frustrating. Two of my team members did not contribute to the project for the first several build days. The other team member was amazing. She helped keep me positive and worked hard.
Because of these delays at the beginning of the project, I had to put in a significant amount of time outside of class. I organised the chemicals, created the reaction cards, and made the slideshow at lunch or after school. I had to devote a lot of time to making sure the presentation night went smoothly.
However, despite the excessive stress that I had because of this project, I am very pleased with the final product. I like the look of the game board and the creativity my team had when designing the game play. I also really liked how we incorporated our LED. I think the lighthouse is very cute and creative.
Because of these delays at the beginning of the project, I had to put in a significant amount of time outside of class. I organised the chemicals, created the reaction cards, and made the slideshow at lunch or after school. I had to devote a lot of time to making sure the presentation night went smoothly.
However, despite the excessive stress that I had because of this project, I am very pleased with the final product. I like the look of the game board and the creativity my team had when designing the game play. I also really liked how we incorporated our LED. I think the lighthouse is very cute and creative.
The Reactions:
-
Reaction 1
-
Reaction 2
-
Reaction 3
-
Catapult
-
Lighthouse
<
>
The first reaction in our game was steel wool and copper chloride. We originally were going to use copper sulfate but since we did not have access to that material, we choose a different solution. In this reaction, Steel wool + copper chloride yields iron sulfate and copper. This is a single replacement reaction.
This reaction is lead nitrate + potassium iodide yields potassium nitrate and lead iodide. It is a double replacement reaction. The lead and the potassium switch partners. A precipitate of lead iodide is formed which is a solid no quite dissolved in a liquid.
The Hydrogen and the sodium are partners with chloride and oxygen. The carbon and the oxygen remain together. Hydrochloric acid and Acetic Acid yield Sodium Chloride, Water, and Carbon Dioxide.
This reaction is acetic acid and sodium bicarbonate also known as baking soda and vinegar. This produces carbon dioxide gas, sodium acetate, and water. When the balloon is filled it pushes up the lever and launches the aluminum foil ball.
|
CH3COOH + NaHCO3 → CH3COONa + CO2 + H20
|
The LED is in our lighthouse and it is off until someone claims the territory it is located in. When someone captures the territory, the player puts copper chloride on the aluminum strip located here. This results in a single replacement reaction. When the reaction is complete the aluminum is now in a solution with the chloride and the copper is floating in it. This stops the circuit from being shorted and the light turns on.